BioLife Solutions Inc.
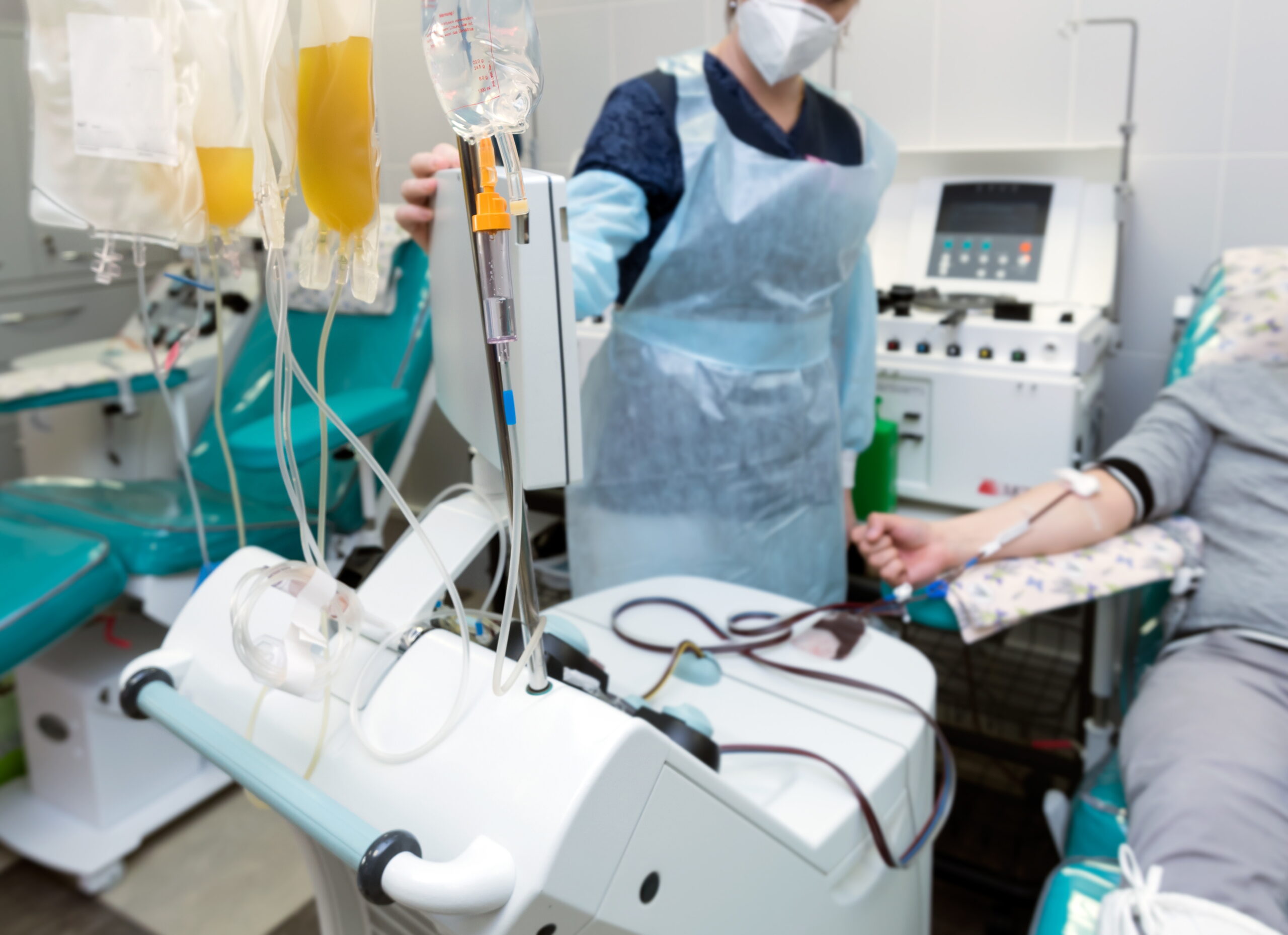
The study data, published in the most recent issue of the journal Cryobiology, showed that BioLife's preservation solutions were able to maintain elevated levels of cell survival throughout the processes used to prepare them for transplantation or cell and tissue engineering, which include freezing, transportation and storage. BioLife's platform technology is based on the Company's unique understanding of the molecular biological and biochemical responses of cells to low temperature exposure, or cryopreservation, which is necessary for preserving cell life.
"Our findings represent a significant breakthrough for the preservation sciences," said BioLife President and CEO John G. Baust, Ph.D., one of the study's authors. "Regenerative medicine and tissue engineering are faced with many hurdles related to product storage and stability, but our new preservation technology gives physicians and scientific researchers solutions for these issues. Additionally, we expect this data will be useful in attracting new customers and development partners and expanding existing relationships."
The data showed that BioLife's CryoStor™ platform, one of the Company's Hypothermosol solutions, extended cell life by 50-100 percent compared to conventional preservation technologies.
A key finding of the study is the ability of the Hypothermosol® solutions to reduce by 50-75 percent or eliminate the need for toxic compounds such as dimethyl sulfoxide (DMSO) in the process, which could be dangerous to patients, said BioLife Vice President of Business Development Robert G. VanBuskirk, Ph.D, another of the study's authors.
"The ability to cryopreserve cells without DMSO could prove to be paramount importance in applications such as bone marrow replacement where the injection into a patient of preserved cellular-based technologies is common," VanBuskirk said.
About BioLife Solutions, Inc.
BioLife Solutions has pioneered the next generation of preservation solutions designed to maintain the viability and health of cellular matter and tissues during freezing, transportation and storage. Based on the Company's proprietary bio-packaging technology and a patented understanding of the mechanism of cellular damage and death, these products enable the biotechnology and medical community to address a growing problem that exists today. The expanding practice of cell and gene therapy has created a need for products that ensure the biological viability of mammalian cell and tissue material during transportation and storage. The HypoThermosol® and CryoStor™ products that the Company is selling today are a significant step forward in meeting these needs.
This news release contains forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995. These forward-looking statements include any statements that relate to the intent, belief, plans or expectations of the Company or its management, or that are not a statement of historical fact. Any forward-looking statements in this news release are based on current expectations and beliefs and are subject to numerous risks and uncertainties that could cause actual results to differ materially. Some of the specific factors that could cause BioLife Solutions' actual results to differ materially are discussed in the Company's recent filings with the Securities and Exchange Commission. BioLife Solutions disclaims any obligation to update any forward-looking statements as a result of developments occurring after the date of this press release.
For further information please contact: investors, Jill Bertotti,
SOURCE: BioLife Solutions Inc.
CONTACT: investors, Jill Bertotti,
Hall,
BioLife Solutions Inc.