BioLife Solutions, Inc.
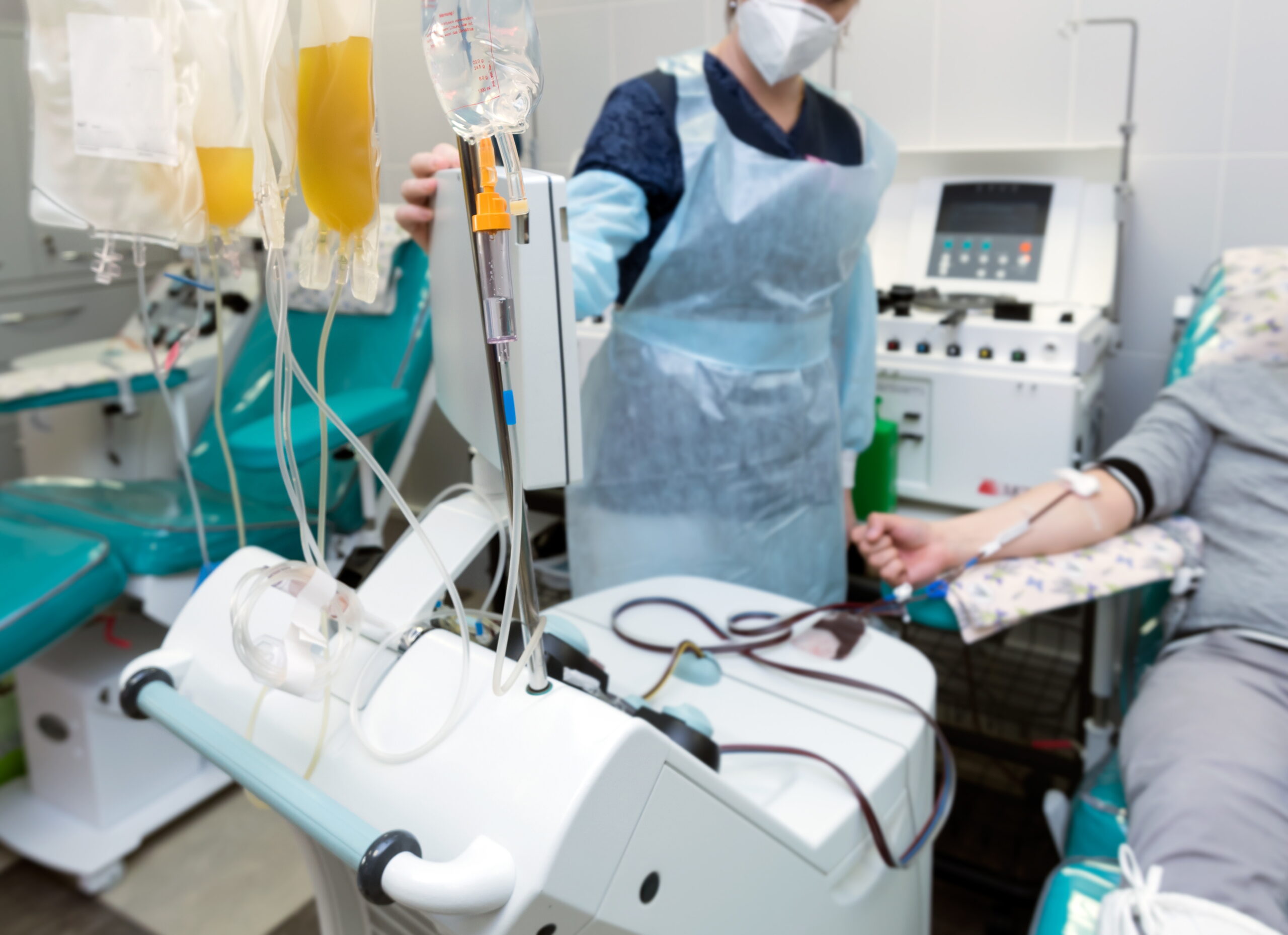
Researchers at the Sydney Cord Blood Bank (Sydney Children's Hospital, NSW, Australia) recognized the limits of traditional cord blood preservation methodologies and conducted a study in which they compared the presently utilized DMSO/Dextran technology with two of BioLife's CryoStor™ cryopreservation products. In all analyses CryoStor™ CS10 proved "significantly superior" to the DMSO/Dextran method. The measures of effectiveness included numbers of nucleated cell recovered, CD34+ cell numbers, CD34+ cell viability and longer-term CFU (clonogenic assay). CFU, an indication of the surviving stem cells' ability to grow and ultimately transform into new cells/tissues, improved 67 percent following preservation in CryoStor™ CS10.
"This study is of fundamental importance to the stem cell community as it provides the first wholly independent comparison of the historic preservation methodology developed in past decades with BioLife's state-of-the-art molecular biological-based preservation products," said John Baust, Ph.D., BioLife's President and Chief Executive Officer. "Much of the stem cell industry is challenged to recognize that older preservation methodologies while long established as a 'gold standard,' offer compromised outcomes. Now, with an independent comparison at hand, cord blood banks have a scientific basis for reevaluation of their preservation processes. Studies such as the one referenced continue to validate the superiority of BioLife's technology."
Cytotherapy is the official journal of the International Society for Cellular Therapy, a leading professional organization in the emerging stem cell-based cell therapy markets. BioLife Solutions, Inc. exhibited at the May 2006 meetings of this Society in Berlin, Germany. The referenced study was titled "Novel Cryoprotectant Significantly Improves the Post-thaw Recovery and Quality of HSB from CB" (Volume 8, No. 1, 57-61, 2006).
About BioLife Solutions
BioLife Solutions, Inc. engages in the development, manufacture, and marketing of cryopreservation solutions, which preserve cells and tissue in low temperature environments for extended periods. It markets HypoThermosol, CryoStor, and GelStor line of solutions directly to companies and labs engaged in pre-clinical research, and to academic institutions. The Company's line of preservation solutions, based on its patented HypoThermosol technology, is composed of synthetic, aqueous solutions containing, in part, minerals and other elements found in human blood, which are necessary to maintain fluids and chemical balances throughout the body at near freezing temperatures. These solutions significantly prolong cell, tissue, and organ viability, which in turn, improve clinical outcomes for new and existing cell, and tissue therapy applications, as well as for organ transplantation. In addition, the Company provides contract-based services for the development of cryopreservation solutions and processes. For more information please visit BioLife Solutions website at www.biolifesolutions.com .
This news release contains forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995. These forward-looking statements include any statements that relate to the intent, belief, plans or expectations of the Company or its management, or that are not a statement of historical fact. Any forward-looking statements in this news release are based on current expectations and beliefs and are subject to numerous risks and uncertainties that could cause actual results to differ materially. Some of the specific factors that could cause BioLife Solutions' actual results to differ materially are discussed in the Company's recent filings with the Securities and Exchange Commission. BioLife Solutions disclaims any obligation to update any forward-looking statements as a result of developments occurring after the date of this press release.
Contact: Allen & Caron Inc
Jill Bertotti (investors)
jill@allencaron.com
Len Hall (media)
len@allencaron.com
949-474-4300
SOURCE: BioLife Solutions, Inc.
CONTACT: Investors, Jill Bertotti,
Hall,
BioLife Solutions, Inc.
Web site: http://www.biolifesolutions.com/